Subscribe to RSS
Please copy the URL and add it into your RSS Feed Reader.
https://www.thieme-connect.de/rss/thieme/en/10.1055-s-00000083.xml
Synlett 2016; 27(13): 2034-2038
DOI: 10.1055/s-0035-1561631
DOI: 10.1055/s-0035-1561631
letter
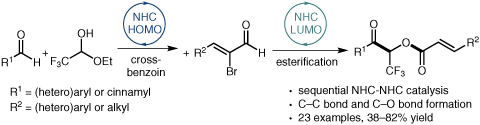
An NHC-Catalyzed Cross-Benzoin–Esterification Sequential Reaction for the Synthesis of Trifluoromethyl-Substituted α,β-Unsaturated Esters
Further Information
Publication History
Received: 02 March 2016
Accepted after revision: 13 April 2016
Publication Date:
03 May 2016 (online)

Abstract
Efficient preparation of synthetically important CF3-containing α,β-unsaturated esters is described using an NHC-catalyzed multicomponent reaction. This approach combines sequential NHC-mediated HOMO and LUMO activation to produce a C–C bond and C–O bond in a one-pot operation.
Key words
organocatalytic reaction - multicomponent reaction - NHC catalysis - trifluoromethylated compounds - cross-benzoin - esterificationSupporting Information
- Supporting information for this article is available online at http://dx.doi.org/10.1055/s-0035-1561631.
- Supporting Information
Primary Data
- for this article are available online at http://www.thieme-connect.com/products/ejournals/journal/10.1055/s-00000083 and can be cited using the following DOI: 10.4125/pd0078th.
- Primary Data
-
References and Notes
- 1 These authors contributed equally.
- 2a Grondal C, Jeanty M, Enders D. Nat. Chem. 2010; 2: 167
- 2b Albrecht Ł, Jiang H, Jørgensen KA. Angew. Chem. Int. Ed. 2011; 50: 8492
- 2c Pellissier H. Adv. Synth. Catal. 2012; 354: 237
- 2d Pellissier H. Tetrahedron 2013; 69: 7171
- 3 Walji AM, MacMillan DW. C. Synlett 2007; 1477
-
4a Enders D, Niemeier O, Henseler A. Chem. Rev. 2007; 107: 5606
- 4b Nair V, Vellalath S, Babu BP. Chem. Soc. Rev. 2008; 37: 2691
- 4c Biju AT, Kuhl N, Glorius F. Acc. Chem. Res. 2011; 44: 1182
- 4d Vora HU, Wheeler P, Rovis T. Adv. Synth. Catal. 2012; 354: 1617
- 4e Févre M, Pinaud J, Gnanou Y, Vignolle J, Taton D. Chem. Soc. Rev. 2013; 42: 2142
- 4f Mahatthananchai J, Bode JW. Acc. Chem. Res. 2014; 47: 696
-
4g Hopkinson MN, Richter C, Schedler M, Glorius F. Nature 2014; 510: 485
-
5 Arduengo AJ, Harlow RL, Kline M. J. Am. Chem. Soc. 1991; 113: 361
- 6a Nair V, Vellalath S, Babu BP. Chem. Soc. Rev. 2008; 37: 2691
-
6b Nair V, Menon RS, Biju AT, Sinu CR, Paul RR, Jose A, Sreekumar V. Chem. Soc. Rev. 2011; 40: 5336
- 7a Moore JL, Rovis T. Top. Curr. Chem. 2009; 291: 118
- 7b Bugaut X, Glorius F. Chem. Soc. Rev. 2012; 41: 3511
- 7c Menon RS, Biju AT, Nair V. Beilstein J. Org. Chem. 2016; 12: 444
- 8a Vora HU, Rovis T. Aldrichimica Acta 2011; 44: 3
-
8b Flanigan DM, Romanov-Michailidis F, White NA, Rovis T. Chem. Rev. 2015; 115: 9307
- 8c Yetra SR, Patra A, Biju AT. Synthesis 2015; 47: 1357
-
9a Sun F.-G, Sun L.-H, Ye S. Adv. Synth. Catal. 2011; 353: 3134
- 9b Zhang B, Feng P, Cui Y, Jiao N. Chem. Commun. 2012; 48: 7280
- 9c Yao C, Jiao W, Xiao Z, Liu R, Li T, Yu C. Tetrahedron 2013; 69: 1133
-
9d Yetra SR, Kaicharla T, Kunte SS, Gonnade RG, Biju AT. Org. Lett. 2013; 15: 5202
- 9e Yetra SR, Roy T, Bhunia A, Porwal D, Biju AT. J. Org. Chem. 2014; 79: 4245
- 9f Jiang D, Dong S, Tang W, Lu T, Du D. J. Org. Chem. 2015; 80: 11593
- 9g Zhang C.-L, Wang D.-L, Chen K.-Q, Ye S. Org. Biomol. Chem. 2015; 13: 11255
- 9h Chen K.-Q, Li Y, Zhang C.-L, Sun D.-Q, Ye S. Org. Biomol. Chem. 2016; 14: 2007
- 9i Xu J, Zhang W, Liu Y, Zhu S, Liu M, Hua X, Chen S, Lu T, Du D. RSC Adv. 2016; 6: 18601
-
10a De Sarkar S, Studer A. Angew. Chem. Int. Ed. 2010; 49: 9266
- 10b Rong Z.-Q, Jia M.-Q, You S.-L. Org. Lett. 2011; 13: 4080
- 10c Biswas A, De Sarkar S, Tebben L, Studer A. Chem. Commun. 2012; 48: 5190
- 10d Samanta RC, De Sarkar S, Fröhlich R, Grimme S, Studer A. Chem. Sci. 2013; 4: 2177
- 10e Bera S, Samanta RC, Daniliuc CG, Studer A. Angew. Chem. Int. Ed. 2014; 53: 9622
- 10f Hu S, Wang B, Zhang Y, Tang W, Fang M, Lu T, Du D. Org. Biomol. Chem. 2015; 13: 4661
- 10g Liang Z.-Q, Wang D.-L, Zhang H.-M, Ye S. Org. Lett. 2015; 17: 5140
- 10h Mao J.-H, Wang Z.-T, Wang Z.-Y, Cheng Y. J. Org. Chem. 2015; 80: 6350
- 10i Yetra SR, Mondal S, Mukherjee S, Gonnade RG, Biju AT. Angew. Chem. Int. Ed. 2016; 55: 268
-
11a Ryan SJ, Candish L, Lupton DW. J. Am. Chem. Soc. 2009; 131: 14176
-
11b Cheng J, Huang Z, Chi YR. Angew. Chem. Int. Ed. 2013; 52: 8592
-
11c Xu J, Jin Z, Chi YR. Org. Lett. 2013; 15: 5028
- 11d Chauhan P, Enders D. Angew. Chem. Int. Ed. 2014; 53: 1485
-
11e Candish L, Levens A, Lupton DW. J. Am. Chem. Soc. 2014; 136: 14397
-
11f Kowalczyk M, Lupton DW. Angew. Chem. Int. Ed. 2014; 53: 5314
- 11g Zhang Z, Zeng X, Xie D, Chen D, Ding L, Wang A, Yang L, Zhong G. Org. Lett. 2015; 17: 5052
-
12a Zeitler K. Org. Lett. 2006; 8: 637
-
12b Kaeobamrung J, Mahatthananchai J, Zheng P, Bode JW. J. Am. Chem. Soc. 2010; 132: 8810
-
12c Zhu Z.-Q, Xiao J.-C. Adv. Synth. Catal. 2010; 352: 2455
-
12d Zhu Z.-Q, Zheng X.-L, Jiang N.-F, Wan X, Xiao J.-C. Chem. Commun. 2011; 47: 8670
- 12e Romanov-Michailidis F, Besnard C, Alexakis A. Org. Lett. 2012; 14: 4906
-
12f Du D, Hu Z, Jin J, Lu Y, Tang W, Wang B, Lu T. Org. Lett. 2012; 14: 1274
- 12g Mahatthananchai J, Kaeobamrung J, Bode JW. ACS Catal. 2012; 2: 494
- 12h Zhou B, Luo Z, Li Y. Chem. Eur. J. 2013; 19: 4428
- 12i Han R, Qi J, Gu J, Ma D, Xie X, She X. ACS Catal. 2013; 3: 2705
- 12j Qi J, Duan X.-Y, Cao L.-L, Wang W.-Y. Asian J. Org. Chem. 2015; 4: 1254
-
13a Ryan SJ, Candish L, Lupton DW. J. Am. Chem. Soc. 2011; 133: 4694
- 13b Ryan S, Candish L, Lupton DW. Synlett 2011; 2275
-
13c Ryan SJ, Stasch A, Paddon-Row MN, Lupton DW. J. Org. Chem. 2012; 77: 1113
-
13d Candish L, Lupton DW. J. Am. Chem. Soc. 2013; 135: 58
- 14 Song X, Ni Q, Grossmann A, Enders D. Chem. Asian J. 2013; 8: 2965
- 15 Ramanjaneyulu BT, Mahesh S, Anand RV. Org. Lett. 2015; 17: 6
- 16a Xie X, Peng C, He G, Leng H.-J, Wang B, Huang W, Han B. Chem. Commun. 2012; 48: 10487
- 16b Li X, Yang L, Peng C, Xie X, Leng H.-J, Wang B, Tang Z.-W, He G, Ouyang L, Huang W, Han B. Chem. Commun. 2013; 49: 8692
- 16c He G, Wu F, Huang W, Zhou R, Ouyang L, Han B. Adv. Synth. Catal. 2014; 356: 2311
- 16d Han B, Xie X, Huang W, Li X, Yang L, Peng C. Adv. Synth. Catal. 2014; 356: 3676
- 16e Han B, Huang W, Ren W, He G, Wang J.-H, Peng C. Adv. Synth. Catal. 2015; 357: 561
- 16f Zhou R, Wu Q, Guo M, Huang W, He X, Yang L, Peng F, He G, Han B. Chem. Commun. 2015; 51: 13113
- 16g Leng H.-J, Peng F, Zingales S, Huang W, Wang B, Zhao Q, Zhou R, He G, Peng C, Han B. Chem. Eur. J. 2015; 21: 18100
- 17a Isanbor C, O'Hagan D. J. Fluorine Chem. 2006; 127: 303
- 17b Müller K, Faeh C, Diederich F. Science 2007; 317: 1881
- 17c Furuya T, Kamlet AS, Ritter T. Nature 2011; 473: 470
- 17d Tomashenko OA, Grushin VV. Chem. Rev. 2011; 111: 4475
- 17e Barata-Vallejo S, Lantaño B, Postigo A. Chem. Eur. J. 2014; 20: 16806
- 17f Bizet V, Besset T, Ma J.-A, Cahard D. Curr. Top. Med. Chem. 2014; 14: 901
- 17g Wang J, Sánchez-Roselló M, Aceña JL, del Pozo C, Sorochinsky AE, Fustero S, Soloshonok VA, Liu H. Chem. Rev. 2014; 114: 2432
- 17h Huang Y.-Y, Yang X, Chen Z, Verpoort F, Shibata N. Chem. Eur. J. 2015; 21: 8664
- 17i Gillis EP, Eastman KJ, Hill MD, Donnelly DJ, Meanwell NA. J. Med. Chem. 2015; 58: 8315
- 18 General Procedure for the Synthesis of CF3-Substituted α,β-Unsaturated Esters The reaction was carried out with aromatic aldehyde 1 (0.5 mmol), CF3CH(OH)OEt (2, 1.0 mmol), precatalyst A (0.05 mmol, 13.7 mg), and DBU (0.1 mmol, 15 μL) in THF (2.0 mL) at 60 °C under argon to afford the acyloin 3, after which α-bromoenal 4 (0.3 mmol) was added in one-pot. The reaction mixture was stirred at 60 °C for a specified reaction time until the reaction completed. Then the reaction mixture was concentrated, and the residue was purified by flash chromatography on silica gel (PE–EtOAc = 40:1) to give the final product.
- 19 Analytical Data for Compound 5a Obtained as a white solid in 65% yield (65.3 mg) for two steps after flash chromatography; mp 58–60 °C. 1H NMR (400 MHz, CDCl3): δ = 8.03–8.01 (m, 2 H), 7.82 (d, J = 16.0 Hz, 2 H), 7.57–7.51 (m, 4 H), 7.44–7.38 (m, 3 H), 6.57 (d, J = 16.0 Hz, 1 H), 6.45 (q, J = 6.8 Hz, 1 H) ppm. 13C NMR (100 MHz, CDCl3): δ = 188.6, 164.8, 148.2, 134.6, 133.7, 131.2, 129.0, 128.9, 128.5, 121.9 (d, JCF = 280 Hz), 115.2, 70.9 (q, JCF = 31 Hz) ppm. ESI-HRMS: m/z calcd for C18H13F3O3 + Na: 357.0714; found: 357.0717.
- 20 Loner CM, Luzzio FA, Demuth DR. Tetrahedron Lett. 2012; 53: 5641
For recent reviews on multicomponent one-pot reactions, see:
For recent reviews on NHC catalysis, see:
For selected reviews on NHC-mediated HOMO activation, see:
For recent reviews on benzoin condensation, see:
For recent reviews on Stetter reaction, see:
For recent reviews on the synthesis of trifluoro-methylated compounds and the applications in medicinal chemistry, see:
